 
That the N-H bonds are polar with a net negative charge on the nitrogenĪtom and a smaller net positive charge on the hydrogen atoms. Of nitrogen (3.04) is greater than that of hydrogen (2.2) the result is Lone pairs can make a contribution to a molecule's dipole moment. Lone pair is a valence e- pair which is not shared with another atom and is sometimes called a non-bonding pair.For ex. Concepts of Physics by HC Verma for JEE.IIT JEE Coaching For Foundation Classes.This all is governed by dipole moment only. You can just imagine there are lone pairs on the central atom carbon which could generate some partial negative character in them and hence the bond between them must not be as strong as the bond between an electropositive and electronegative atom. In most of the cases the central atom in any compound of oxygen is more electropositive atom and electropositive atoms do not contain lone pairs. These electrons are a part of oxygen and these makes oxygen more electronegative because of which the dipole moment increases ans hence the bond becomes stronger. The lone pair is generally resided on the other side of the bond. Coming to the topic of dipole moments you should consider the lone pair of electrons on oxygen. Here the formal charge on carbon is zero and the same formal charge is there on both the oxygens. It can cause hindrances by charge imbalances which could result in imbalance of charges and which could destabilise the atom due to decrease in dipole moment.Ĭonsider a molecule of carbon dioxide where two oxygens are present at each side of carbon: The dipole moment is actually affected by the presence of a lone pair of electrons because the electrons on the central atom can cause shielding effect as the inner orbital electrons does for the outer-orbitals. The first order term (or the second term) of the multipole expansion of a function.Topological dipole moment, the measure of the topological defect charge distribution.Nuclear magnetic moment, the magnetic moment of an atomic nucleus.Magnetic dipole moment, the measure of the magnetic polarity of a system of charges.Electron electric dipole moment, the measure of the charge distribution within an electron.Bond dipole moment, the measure of polarity of a chemical bond.Molecular dipole moment, the electric dipole moment of a molecule.Transition dipole moment, the electrical dipole moment in quantum mechanics.Electric dipole moment, the measure of the electrical polarity of a system of charges. 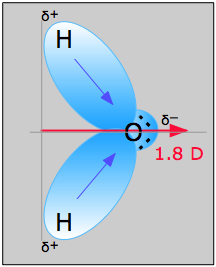
But, the directionality (or lack thereof) of the lone pair must also be assessed, since a lack of directionality may preclude it from making a significant contribution to the overall molecular dipole moment.ĭipole moment can be defined as the product of magnitude of charges and the distance of separation between the charges. So in summary, a lone pair of electrons can make a significant contribution to the magnitude of a molecular dipole moment due to the fact that they are more localized than bonding electrons and consequently there is a high electron density. You can see that in this case, the lone pair on phosphorous, due to its spherical symmetry will not make a contribution to the overall molecular dipole moment. The molecules appear to be very similar, they are in the same column in the Periodic Table. Consider the two molecules pictured below, ammonia and phosphine. Next we must understand the directionality of the lone pair of electrons. This increased electron density could lead to a more significant contribution from the lone pair electrons to the molecular dipole moment than from electrons spread out more diffusely in a covalent bond. Unlike a typical covalent bond where the electrons are shared between two nuclei and the electron density is spread out over the entire bond, in a lone pair the electrons are not shared and the electron density is more localized around the atom that has the lone pair of electrons. There is no single answer to your question, let me explain. How does lone pair of a central atom affect the dipole moment? 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
